Category: Laboratory Calculations
Ponceau S (Tetrasodium Salt) [C22H12N4Na4O13S4] Molecular Weight Calculation
Ponceau S (tetrasodium salt) (C22H12N4Na4O13S4) is an organic compound of six elements: Carbon, Hydrogen, Nitrogen, Sodium, Oxygen, and Sulfur. The molecular weight of Ponceau S (tetrasodium salt) is 760.5736371 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Molarity of 25% (w/w) Hydrochloric Acid (HCl)
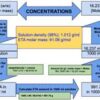
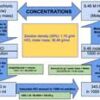
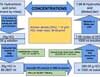
The molarity of 25% (w/w) hydrochloric acid is 7.68 M. The 25% (w/w) hydrochloric acid contains 25g of HCl per 100g of solution. Since molarity is the number of moles of a substance present in a liter of solution, one must first calculate how much HCl is present in 1L of 25% hydrochloric acid solution. Once we know the amount of HCl present in 1L of solution, we can calculate the molarity of the solution by dividing the HCl amount by the HCl molecular weight.
2,6-Dichloropyridine-3-carboxaldehyde (C6H3Cl2NO) Molecular Weight Calculation
2,6-Dichloropyridine-3-carboxaldehyde (C6H3Cl2NO) is an organic compound of five elements: Carbon, Hydrogen, Chlorine, Nitrogen, and Oxygen. The molecular weight of 2,6-Dichloropyridine-3-carboxaldehyde is 175.99974 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.

![3,5-Diphenyl-isoxazole [C15H11NO] Molecular Weight Calculation 3,5-Diphenyl-isoxazole [C15H11NO] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/10/fi-35-diphenyl-isoxazole-molecular-weight-calculation.jpg)
![Thiazole [C3H3NS] Molecular Weight Calculation Thiazole [C3H3NS] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/10/fi-thiazole-molecular-weight-calculation.jpg)
![Isoxazole [C3H3NO] Molecular Weight Calculation Isoxazole [C3H3NO] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/10/fi-isoxazole-molecular-weight-calculation.jpg)
![Ponceau S (Tetrasodium Salt) [C22H12N4Na4O13S4] Molecular Weight Calculation Ponceau S (tetrasodium salt) [C22H12N4Na4O13S4] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/10/fi-ponceau-s-tetrasodium-salt-molecular-weight-calculation-100x92.jpg)
![Benzothiazole [C7H5NS] Molecular Weight Calculation Benzoxazole [C7H3Cl2NS] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/11/fi-benzoxazole-molecular-weight-calculation-100x68.jpg)
![2,6-Dichloropyridine-3-carboxaldehyde (C6H3Cl2NO) Molecular Weight Calculation 2,6-Dichloropyridine-3-carboxaldehyde [C6H3Cl2NO] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/09/fi-26-dichloropyridine-3-carboxaldehyde-molecular-weight-calculation.jpg)