![]()
The molecular weight of Sodium chloride (NaCl) is 58.44246928.
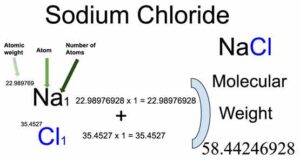 Sodium chloride (NaCl) is an inorganic compound of two elements: Sodium and Chlorine. The molecular weight of Sodium chloride is 58.44246928 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of Sodium and Chlorine.
Sodium chloride (NaCl) is an inorganic compound of two elements: Sodium and Chlorine. The molecular weight of Sodium chloride is 58.44246928 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of Sodium and Chlorine.
CALCULATION PROCEDURE: Sodium Chloride (NaCl) Molecular Weight Calculation
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Sodium chloride molecule.
From the chemical formula, you will know different atoms and their number in a Sodium chloride molecule. The chemical formula of Sodium chloride is NaCl. From the chemical formula, you can find that one molecule of Sodium chloride has one Sodium (Na) and one Chlorine (Cl) atom.
Step 2: Find out the atomic weights of each atom (from the periodic table).
Atomic weight of Sodium (Na): 22.98976928 (Ref: Ciaaw-sodium)
Atomic weight of Chlorine (Cl): 35.4527 (Ref: Jlab-ele017)
Step 3: Calculate the total weight of each atom present in Sodium chloride by multiplying its atomic weight by its number.
Number of Sodium atoms in Sodium chloride: 1
Atomic weight of Sodium: 22.98976928
Total weight of Sodium atoms in Sodium chloride: 22.98976928
Number of chlorine atoms in hydrogen chloride: 1
Atomic weight of chlorine: 35.4527
Total weight of chlorine atoms in Sodium chloride: 35.4527
Step 4: Calculate the molecular weight of Sodium chloride by adding up the total weight of all atoms.
Molecular weight of Sodium chloride (NaCl): 22.98976928 (Sodium) + 35.4527 (chlorine) = 58.44246928
So the molecular weight of Sodium chloride is 58.44246928.
Sodium Chloride (NaCl) Molecular Weight Calculation
| Molecular weight of Sodium Chloride (NaCl) | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Hydrogen (H) | 1 | 22.98976928 | 22.98976928 |
| Chlorine (Cl) | 1 | 35.4527 | 35.4527 |
| Molecular weight of Sodium chloride (NaCl): | 58.44246928 | ||
REFERENCES:
- Ciaaw-sodium: https://www.ciaaw.org/sodium.htm
- Jlab-ele017: https://education.jlab.org/itselemental/ele017.html