Tag: hydrogen chloride
Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation
![]()
Tris (C4H11NO3) is an organic compound of four elements: Carbon, Hydrogen, Nitrogen, and Oxygen. The Tris hydrochloride (C4H11NO3.HCl) also contains one molecule of Hydrogen chloride. The molecular weight of Tris hydrochloride is 157.59644 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Preparation of 1M Hydrochloric Acid From Concentrated Stock Solution (37%, w/w)
![]()
A 37% (w/w) hydrochloric acid can be purchased from several suppliers. It is a clear colorless liquid and can be diluted to prepare solutions of known concentrations. Remember that the molarity of hydrochloric acid is equal to the normality of the solution, which means 1M solution is also a 1N solution. Here we describe a procedure to prepare 1M hydrochloric acid solution by diluting a 37% (w/w) concentrated hydrochloric acid solution.
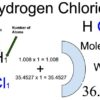
Hydrogen Chloride (HCl) Molecular Weight Calculation
![]()
Hydrogen chloride (HCl) is an inorganic compound of two elements: hydrogen and chlorine. The hydrogen chloride molecular weight is the sum of hydrogen and chlorine atomic weights.
Hydrochloric Acid
![]()
Aqueous solution of hydrogen chloride (HCl) gas is called hydrochloric acid. Hydrochloric acid is a very strong acid. It hydrolyzes in water into H+ and Cl﹣. The pKa of hydrochloric acid is -6.0.
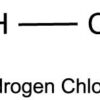
Hydrogen Chloride (HCl)
![]()
Hydrogen chloride (HCl) is an inorganic compound of two elements: hydrogen (atomic number 1) and chlorine (atomic number 17). Hydrogen chloride is a colourless to yellowish, corrosive, nonflammable gas at room temperature. The gas is heavier than air and has a strong irritating odor. It is readily soluble in water and forms an acidic solution. Its solution in water is called hydrochloric acid. Hydrochloric acid is a very strong acid. It hydrolyzes in water into H+ (H3O+) and Cl﹣.
Molarity of 37% (w/w) Hydrochloric Acid (HCl)
![]()
A 37% (w/w) concentrated Hydrochloric acid is a clear colorless liquid. It is an aqueous solution of hydrogen chlori (HCl). The 100 g of 37% (w/w) Hydrochloric acid contains 37 g of HCl. To calculate the molarity, one must first calculate how much HCl is present in 1 L of 37% Hydrochloric acid solution. Once we know the amount of HCl present in 1 L solution, we can calculate the molarity of the solution by dividing the HCl amount by the HCl molecular weight.
![Tris Hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation Tris hydrochloride [C4H11NO3.HCl] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/11/fi-tris-hydrochloride-molecular-weight-calculation-100x87.jpg)