![]()
The molecular weight of Sodium orthovanadate (Na3VO4) is 183.90840784.
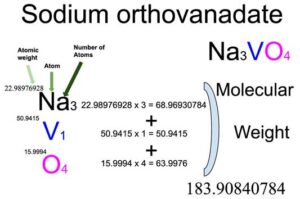 Sodium orthovanadate (Na3VO4) is an inorganic compound of three elements: Sodium, Vanadium, and Oxygen. The molecular weight of Sodium orthovanadate is 183.90840784 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Sodium orthovanadate (Na3VO4) is an inorganic compound of three elements: Sodium, Vanadium, and Oxygen. The molecular weight of Sodium orthovanadate is 183.90840784 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
CALCULATION PROCEDURE: Sodium orthovanadate [Na3VO4] Molecular Weight Calculation
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Sodium orthovanadate molecule.
You will know different atoms and their number in a Sodium orthovanadate molecule from the chemical formula. The chemical formula of Sodium orthovanadate is Na3VO4. From the chemical formula, you can find that one molecule of Sodium orthovanadate has three Sodium (Na) atoms, one Vanadium (V) atom, and four Oxygen (O) atoms.
Step 2: Find out the atomic weights of each atom (from the periodic table).
Atomic weight of Sodium (Na): 22.98976928 (Ref: Ciaaw-sodium)
Atomic weight of Vanadium (V): 50.9415 (Ref: Jlab-ele023)
Atomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008)
Step 3: Calculate the molecular weight of Sodium orthovanadate by adding the total weight of all atoms.
Number of Sodium (Na) atoms in Sodium orthovanadate: 3
Atomic weight of Sodium: 22.98976928
Total weight of Sodium atoms in Sodium orthovanadate: 22.98976928 x 3 = 68.96930784
Number of Vanadium (V) atoms in Sodium orthovanadate: 1
Atomic weight of Vanadium: 50.9415
Total weight of Vanadium atoms in Sodium orthovanadate: 50.9415 x 1 = 50.9415
Number of Oxygen (O) atoms in Sodium orthovanadate: 4
Atomic weight of Oxygen: 15.9994
Total weight of Oxygen atoms in Sodium orthovanadate: 15.9994 x 4 = 63.9976
Step 4: Calculate the molecular weight of Sodium orthovanadate by adding up the total weight of all atoms.
Molecular weight of Sodium orthovanadate : 68.96930784 (Sodium) + 50.9415 (Vanadium) + 63.9976 (Oxygen) = 183.90840784
So the molecular weight of Sodium orthovanadate is 183.90840784.
Sodium orthovanadate [Na3VO4] Molecular Weight Calculation
| Molecular weight of Sodium orthovanadate (Na3VO4) | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Sodium (Na) | 3 | 22.98976928 | 68.96930784 |
| Vanadium (V) | 1 | 50.9415 | 50.9415 |
| Oxygen (O) | 4 | 15.9994 | 63.9976 |
| Molecular weight of Sodium orthovanadate (Na3VO4): | 183.90840784 | ||
REFERENCES:
- Ciaaw-sodium : https://www.ciaaw.org/sodium.htm
- Jlab-ele023: https://education.jlab.org/itselemental/ele023.html
- Jlab-ele008: https://education.jlab.org/itselemental/ele008.html
RELATED POSTS
Article authenticity Index: **
* (Newly added)
***** (Revised 4 times)
********** (Revised 9 times)
# Please let us know if you find any mistakes. Your comments are welcome in the comment box.