![]()
The molecular weight of Trimethylene carbonate (C4H6O3) is 102.089.
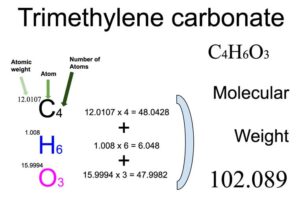 To calculate molecular weight of any compound, the first step is to know the constituent atoms and their number in that particular compound. Then calculate the total weight of each atom by multiplying its atomic weight by its number. The sum of total weight of all constituent atoms will be the molecular weight of the compound. Note that the value of atomic weight may differ slightly from different sources.
To calculate molecular weight of any compound, the first step is to know the constituent atoms and their number in that particular compound. Then calculate the total weight of each atom by multiplying its atomic weight by its number. The sum of total weight of all constituent atoms will be the molecular weight of the compound. Note that the value of atomic weight may differ slightly from different sources.
CALCULATION PROCEDURE: Trimethylene carbonate (C4H6O3) Molecular Weight Calculation
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Trimethylene carbonate molecule.
From the chemical formula, you will know different atoms and their number in a Trimethylene carbonate molecule. Chemical formula of Trimethylene carbonate is C4H6O3. From the chemical formula of Trimethylene carbonate, you can find that one molecule of Trimethylene carbonate has four Carbon (C) atoms, six Hydrogen (H) atoms and three Oxygen (O) atoms.
Step 2: Find out atomic weights of each atom (from periodic table).
Atomic weight of Carbon (C): 12.0107 (Ref: Jlab-ele006)
Atomic weight of Hydrogen (H) : 1.008 (Ref: Lanl-1)
Atomic weight of Oxygen (O) : 15.9994 (Ref: Jlab-ele008)
Step 3: Calculate the total weight of each atom present in a Trimethylene carbonate molecule by multiplying its atomic weight by its number.
Number of Carbon atoms in Trimethylene carbonate: 4
Atomic weight of Carbon: 12.0107
Total weight of Carbon atoms in Trimethylene carbonate: 12.0107 x 4 = 48.0428
Number of Hydrogen atoms in Trimethylene carbonate: 6
Atomic weight of Hydrogen: 1.008
Total weight of Hydrogen atoms in Trimethylene carbonate: 1.008 x 6 = 6.048
Number of Oxygen atoms in Trimethylene carbonate: 3
Atomic weight of Oxygen: 15.9994
Total weight of Oxygen atoms in Trimethylene carbonate: 15.9994 x 3 = 47.9982
Step 4: Calculate the molecular weight of Trimethylene carbonate by adding up the total weight of all atoms.
Molecular weight of Trimethylene carbonate: 48.0428 (Carbon) + 6.048 (Hydrogen) + 47.9982 (Oxygen) = 102.089
So the molecular weight of Trimethylene carbonate is 102.089.
Trimethylene carbonate (C4H6O3) Molecular Weight Calculation
| Molecular weight of Trimethylene carbonate | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Carbon (C) | 4 | 12.0107 | 48.0428 |
| Hydrogen (H) | 6 | 1.008 | 6.048 |
| Oxygen (O) | 3 | 15.9994 | 47.9982 |
| Molecular weight of Trimethylene carbonate: | 102.089 | ||
REFERENCES:
- Lanl-1: https://periodic.lanl.gov/1.shtml
- Pubchem-Hydrogen : https://pubchem.ncbi.nlm.nih.gov/element/Hydrogen
- Jlab-ele006; https://education.jlab.org/itselemental/ele006.html
- Pubchem-6: https://pubchem.ncbi.nlm.nih.gov/element/6
- Jlab-ele008: https://education.jlab.org/itselemental/ele008.html
- Pubchem-8: https://pubchem.ncbi.nlm.nih.gov/element/8