![]()
The molecular weight of Magnesium Chloride (MgCl2) is 95.2104.
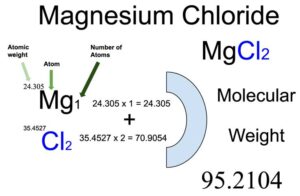 Magnesium chloride (MgCl2) is an organic compound of two elements: Magnesium and Chlorine. The molecular weight of Magnesium chloride is 95.2104 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Magnesium chloride (MgCl2) is an organic compound of two elements: Magnesium and Chlorine. The molecular weight of Magnesium chloride is 95.2104 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
CALCULATION PROCEDURE: Magnesium Chloride (MgCl2) Molecular Weight Calculation
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Magnesium chloride molecule.
You will know different atoms and their number in a Magnesium chloride molecule from the chemical formula. The chemical formula of Magnesium chloride is MgCl2. The chemical formula shows that one molecule of Magnesium chloride consists of one Magnesium (Mg) atom and two Chlorine (Cl) atoms.
Step 2: Find out the atomic weights of each atom.
Atomic weight of magnesium (Mg): 24.305 (Ref: Pubchem-12, Jlab-ele012)
Atomic weight of chlorine (Cl): 35.4527 (Ref: Pubchem-Chlorine, jlab-ele017)
Step 3: Calculate the molecular weight of Magnesium chloride by adding the total weight of all atoms.
Number of magnesium atoms in Magnesium chloride: 1
Atomic weight of magnesium: 24.305
Total weight of magnesium atoms in Magnesium chloride: 24.305 x 1 = 24.305
Number of chlorine atoms in Magnesium chloride: 2
Atomic weight of chlorine: 35.4527
Total weight of chlorine atoms in Magnesium chloride: 35.4527 x 2 = 70.9052
Step 4: Calculate the molecular weight of Magnesium chloride by adding up the total weight of all atoms.
Molecular weight of Magnesium chloride (MgCl2): 24.305 (Magnesium) + 70.90 (Chlorine) = 95.2104
So the molecular weight of Magnesium chloride is 95.2104.
Magnesium Chloride (MgCl2) Molecular Weight Calculation
| Molecular weight of Magnesium chloride (MgCl2) | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Mg (Magnesium) | 1 | 24.305 | 24.305 |
| Cl (Chlorine) | 2 | 35.4527 | 70.9052 |
| Molecular weight of Magnesium chloride (MgCl2): | 95.2104 | ||
REFERENCES:
- Pubchem-12: https://pubchem.ncbi.nlm.nih.gov/element/12
- Jlab-ele012: https://education.jlab.org/itselemental/ele012.html
- Pubchem-Chlorine: https://pubchem.ncbi.nlm.nih.gov/element/Chlorine
- jlab-ele017: https://education.jlab.org/itselemental/ele017.html