![]()
The molecular weight of MOPS (4-Morpholinepropanesulfonic acid) [C7H15NO4S] is 209.26524.
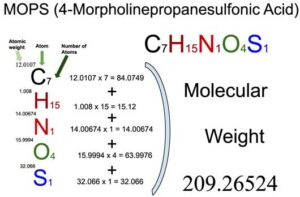 MOPS (4-Morpholinepropanesulfonic acid) (C7H15NO4S) is an organic compound of five elements: Carbon, Hydrogen, Nitrogen, Oxygen, and Sulfur. The molecular weight of MOPS (4-Morpholinepropanesulfonic acid) is 209.26524 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
MOPS (4-Morpholinepropanesulfonic acid) (C7H15NO4S) is an organic compound of five elements: Carbon, Hydrogen, Nitrogen, Oxygen, and Sulfur. The molecular weight of MOPS (4-Morpholinepropanesulfonic acid) is 209.26524 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
CALCULATION PROCEDURE: MOPS (4-Morpholinepropanesulfonic acid) [C7H15NO4S] Molecular Weight Calculation
Step 1: Find the chemical formula and determine constituent atoms and their number in a MOPS (4-Morpholinepropanesulfonic acid) molecule.
You will know different atoms and their number in a MOPS (4-Morpholinepropanesulfonic acid) molecule from the chemical formula. The chemical formula of MOPS (4-Morpholinepropanesulfonic acid) is C7H15NO4S. From the chemical formula, you can find that one molecule of MOPS (4-Morpholinepropanesulfonic acid) consists of seven Carbon (C) atoms, fifteen Hydrogen (H) atoms, one Nitrogen (N) atom, four Oxygen atoms, and one Sulfur (S) atom.
Step 2: Find out the atomic weights of each atom (from the periodic table).
Atomic weight of Carbon (C): 12.0107 (Ref: Jlab-ele006)
Atomic weight of Hydrogen (H): 1.008 (Ref: Lanl-1)
Atomic weight of Nitrogen (N): 14.00674 (Ref: Jlab-ele007)
Atomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008)
Atomic weight of Sulfur (S): 32.066 (Ref: Jlab-ele016)
Step 3: Calculate the total weight of each atom in a MOPS (4-Morpholinepropanesulfonic acid) molecule by multiplying its atomic weight by its number.
Number of Carbon atoms in MOPS (4-Morpholinepropanesulfonic acid): 7
Atomic weight of Carbon (C): 12.0107
Total weight of Carbon atoms in MOPS (4-Morpholinepropanesulfonic acid): 12.0107 x 7 = 84.0749
Number of Hydrogen atoms in MOPS (4-Morpholinepropanesulfonic acid): 15
Atomic weight of Hydrogen (H): 1.008
Total weight of Hydrogen atoms in MOPS (4-Morpholinepropanesulfonic acid): 1.008 x 15 = 15.12
Number of Nitrogen atoms in MOPS (4-Morpholinepropanesulfonic acid): 1
Atomic weight of Nitrogen (N): 14.00674
Total weight of Nitrogen atoms in MOPS (4-Morpholinepropanesulfonic acid): 14.00674 x 1 = 14.00674
Number of Oxygen atoms in MOPS (4-Morpholinepropanesulfonic acid): 4
Atomic weight of Oxygen (O): 15.9994
Total weight of Oxygen atoms in MOPS (4-Morpholinepropanesulfonic acid): 15.9994 x 4 = 63.9976
Number of Sulfur atoms in MOPS (4-Morpholinepropanesulfonic acid): 1
Atomic weight of Sulfur (S): 32.066
Total weight of Sulfur atoms in MOPS (4-Morpholinepropanesulfonic acid): 32.066 x 1 = 32.066
Step 4: Calculate the molecular weight of MOPS (4-Morpholinepropanesulfonic acid) by adding up the total weight of all atoms.
Molecular weight of MOPS (4-Morpholinepropanesulfonic acid): 84.0749 (Carbon) + 15.12 (Hydrogen) + 14.00674 (Nitrogen) + 63.9976 (Oxygen) + 32.066 (Sulfur) = 209.26524
So the molecular weight of MOPS (4-Morpholinepropanesulfonic acid) is 209.26524.
MOPS (4-Morpholinepropanesulfonic acid) [C7H15NO4S] Molecular Weight Calculation
| Molecular weight of MOPS (4-Morpholinepropanesulfonic acid) [C7H15NO4S] | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Carbon (C) | 7 | 12.0107 | 84.0749 |
| Hydrogen (H) | 15 | 1.008 | 15.12 |
| Nitrogen (N) | 1 | 14.00674 | 14.00674 |
| Oxygen (O) | 4 | 15.9994 | 63.9976 |
| Sulfur (S) | 1 | 32.066 | 32.066 |
| Molecular weight of MOPS (4-Morpholinepropanesulfonic acid) [C7H15NO4S]: | 209.26524 | ||
REFERENCES:
- Lanl-1: https://periodic.lanl.gov/1.shtml
- Jlab-ele006: https://education.jlab.org/itselemental/ele006.html
- Jlab-ele007: https://education.jlab.org/itselemental/ele007.html
- Jlab-ele008: https://education.jlab.org/itselemental/ele008.html
- Jlab-ele016: https://education.jlab.org/itselemental/ele016.html