![]()
OVERVIEW
- A 37% (w/w) Hydrochloric Acid is a clear colorless aqueous solution of Hydrogen chloride (HCl) gas. It can be purchased from several commercial suppliers.
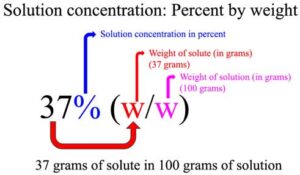
- The “%” refers to solution concentration in percent and “(w/w)” refers to solute and solution amount given in grams (i.e., percentage by weight). This means a 37% (w/w) Hydrochloric acid contains 37 g of HCl per 100 g of solution.
- The density of 37% (w/w) Hydrochloric acid solution is 1.2 g/ml at 25° C. This means the weight of the 1 ml of the hydrochloric acid solution is 1.2 g at 25°C.
- Molarity refers to the number of moles of the solute present in 1 liter of solution. To calculate the Molarity of 37% (w/w) Hydrochloric Acid (HCl), you need to know how many moles of HCl are present in 1 liter of solution.
- In simple words, 1 mole is equal to the molecular weight of the substance. For example, 1 mole of Hydrogen chloride is equal to 36.46 g of hydrogen chloride (Molecular Weight: 36.46).
- To calculate the molarity, one must first calculate how much Hydrogen chloride is present in 1 L of 37% Hydrochloric acid. Once we know the amount of HCl present in 1 L solution, we can calculate the molarity of the solution by dividing the HCl amount by the molecular weight.

RELATED POSTS
- Calculator: Converting Percentage by Mass to Molarity
- Molarity of 25% (w/w) Hydrochloric Acid (HCl)
- Molarity of 30% (w/w) Hydrochloric Acid (HCl)
- Molarity of 32% (w/w) Hydrochloric Acid (HCl)
- Molarity of 37% (w/w) Hydrochloric Acid (HCl)
- How to Calculate Molarity of 37% (w/w) Hydrochloric acid (HCl), Alternative Method

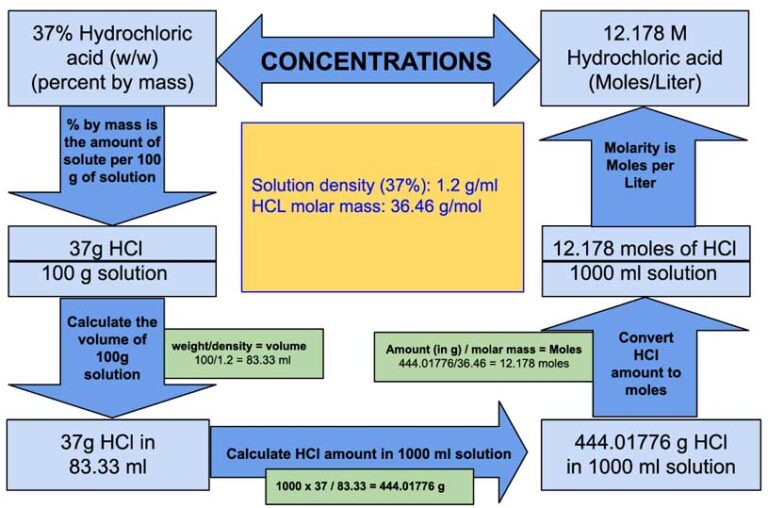
Calculation procedure
| Known values | |
| Molar mass of Hydrogen chloride | 36.46 g/mole |
| Concentration of Hydrochloric acid | 37% (% by mass, w/w) |
| Density of 37% Hydrochloric acid | 1.2 g/ml |
Step 1: Calculate the volume of 100 grams of Hydrochloric acid.
Formula:
Density =
OR
Volume =
The volume of 100 g of Hydrochloric acid : = 83.33 ml
Note: 37% (w/w) Hydrochloric acid means that 100 g of Hydrochloric acid contains 37 g of HCl.
The volume of 100 g of Hydrochloric acid is 83.33 ml which means 37 g of HCl is present in 83.33 ml of Hydrochloric acid.
Step 2: Calculate how many grams of HCl is present in 1000 ml of Hydrochloric acid.
83.33 ml of Hydrochloric acid contains = 37 grams of HCl
1 ml of Hydrochloric acid will contain = grams of HCl
1000 ml of Hydrochloric acid will contain = grams of HCl = 444.01776 grams of HCl
1000 ml of Hydrochloric acid will contain 444.01776 grams of HCl.
Step 3: Calculate the number of moles of HCl in 444.01776 grams of HCl.
36.46 g of HCl is equal to 1 mole.
1 g of HCl will be equal to moles.
444.01776 grams of Hydrogen chloride will be equal to = = 12.178 moles
Therefore, we can say that 1 liter of Hydrochloric acid contains 12.178 moles of HCl or in other words molarity of 37% (w/w) Hydrochloric acid is equal to 12.178 M.
Calculator – Calculate the molarity of concentrated Hydrochloric acid (HCl)
Hydrogen Chloride (HCl) Molecular weight: 36.46 g/mole
(Change the % (w/w) concentration)
Density of Hydrochloric acid: g/ml
(Change the density)
Molarity of Hydrochloric acid: 12.178 M
I think the calculation above is wrong. I think the proper answer is as follows:
37w/w% means that of 1000g of solution 370g is HCl. 370g / 36.46g = 10.15M
ok wait now its clear to me:
1L of 37w/w% HCl weighs 1200g
37w% of 1200g is 444g
444g / 36.5g = 12.2M