The molecular weight of Calcium chloride tetrahydrate [CaCl2.4H2O] (CAS Registry Number: 25094-02-4) is 183.045.
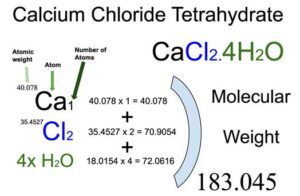 Calcium Chloride (CaCl2) is an inorganic compound of two elements: Calcium (Ca) and Chlorine (Cl). The tetrahydrate form of Calcium Chloride (CaCl2.4H2O) also contains 4 water molecules. The molecular weight of Calcium chloride tetrahydrate is 183.045 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements and water molecules.
Calcium Chloride (CaCl2) is an inorganic compound of two elements: Calcium (Ca) and Chlorine (Cl). The tetrahydrate form of Calcium Chloride (CaCl2.4H2O) also contains 4 water molecules. The molecular weight of Calcium chloride tetrahydrate is 183.045 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements and water molecules.
CALCULATION PROCEDURE: Calcium chloride tetrahydrate (CaCl2.4H2O) Molecular Weight Calculation
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Calcium chloride tetrahydrate molecule.
You will know different atoms and their number in a Calcium chloride tetrahydrate molecule from the chemical formula. The chemical formula of Calcium chloride tetrahydrate is CaCl2.4H2O. From the chemical formula, you can find that one molecule of Calcium chloride tetrahydrate consists of one Calcium (Ca) atom, two Chlorine (Cl) atoms, and four molecules of water.
Step 2: Find out the atomic weights of each atom (from the periodic table).
Atomic weight of Calcium (Ca): 40.078 (Ref: Ciaaw-calcium, Jlab-ele020)
Atomic weight of Chlorine (Cl): 35.4527 (Ref: Jlab-ele017)
Molecular weight of Water [H2O]: 18.0154
Step 3: Calculate the molecular weight of Calcium chloride tetrahydrate by adding the total weight of all atoms.
Number of Calcium atoms in Calcium chloride tetrahydrate: 1
Atomic weight of Calcium: 40.078
Total weight of Calcium atoms in Calcium chloride tetrahydrate: 40.078 x 1 = 40.078
Number of Chlorine atoms in Calcium chloride tetrahydrate: 2
Atomic weight of Chlorine: 35.4527
Total weight of Chlorine atoms in Calcium chloride tetrahydrate: 35.4527 x 2 = 70.9054
Number of water (H2O) molecules in Calcium chloride tetrahydrate: 4
Molecular weight of water: 18.0154
Total weight of water in Calcium chloride tetrahydrate: 18.0154 x 4 = 72.0616
Step 4: Calculate the molecular weight of Calcium chloride tetrahydrate by adding up the total weight of all atoms.
Molecular weight of Calcium chloride tetrahydrate (CaCl2.6H2O): 40.078 (Calcium) + 70.9054 (Chlorine) + 72.0616 (water) = 183.045
So the molecular weight of Calcium chloride tetrahydrate is 183.045.
Calcium Chloride Tetrahydrate (CaCl2.4H2O) Molecular Weight Calculation
| Molecular weight of Calcium chloride Tetrahydrate (CaCl2.4H2O) | |||
| Constituent atoms | Number of each atom | Atomic weight | Total weight |
| Calcium (Ca) | 1 | 40.076 | 40.078 |
| Chlorine (Cl) | 2 | 35.4527 | 70.9054 |
| Water (H2O) | 4 | 18.0154 | 72.0616 |
| Molecular weight of Calcium chloride tetrahydrate (CaCl2.4H2OC): | 183.045 | ||
REFERENCES:
- Ciaaw-calcium: https://www.ciaaw.org/calcium.htm
- Jlab-ele020: https://education.jlab.org/itselemental/ele020.html
- jlab-ele017: https://education.jlab.org/itselemental/ele017.html
RELATED POSTS
- Calcium Chloride (CaCl2) Molecular Weight Calculation
- Calcium Chloride Dihydrate (CaCl2.2H2O) Molecular Weight Calculation
- Calcium Chloride Tetrahydrate (CaCl2.4H2O) Molecular Weight Calculation
- Calcium Chloride Hexahydrate (CaCl2.6H2O) Molecular Weight Calculation
FURTHER READING
- PubChem Calcium chloride tetrahydrate
- ChemSpider Calcium chloride tetrahydrate
Article authenticity Index: ***
* (Newly added)
***** (Revised 4 times)
********** (Revised 9 times)
# Please let us know if you find any mistakes. Your comments are welcome in the comment box.
4 thoughts on “Calcium Chloride Tetrahydrate (CaCl2.4H2O) Molecular Weight Calculation”