Author: admin
Suppliers: Sodium Phosphate Dibasic (Na2HPO4)
![]()
Note: Sodium Phosphate Dibasic (Na2HPO4) can be purchased from the following suppliers. You must visit the supplier’s product page to…
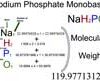
Sodium Phosphate Monobasic (NaH2PO4) Molecular Weight Calculation
![]()
The molecular weight of Sodium phosphate monobasic [NaH2PO4] is 119.977131278. Sodium phosphate monobasic (NaH2PO4), also called Monosodium phosphate or monobasic…
Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation
![]()
Sodium phosphate dibasic (Na2HPO4), also called Disodium phosphate (DSP) or disodium hydrogen phosphate, is an inorganic compound of four elements: Sodium, Hydrogen, Phosphorus, and Oxygen. The dihydrate form of Sodium phosphate dibasic (Na2HPO4.2H2O) also contains 2 water molecules. The molecular weight of Sodium phosphate dibasic dihydrate is 177.9897 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements and water molecules.
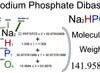
Sodium Phosphate Dibasic (Na2HPO4) Molecular Weight Calculation
![]()
Sodium phosphate dibasic (Na2HPO4) is an inorganic compound of four elements: Sodium, Hydrogen, Phosphorus, and Oxygen. The molecular weight of Sodium phosphate dibasic is 141.9589 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Glutaraldehyde [(CH2)3(CHO)2] Molecular Weight Calculation
![]()
Glutaraldehyde [(CH2)3(CHO)2] is an organic compound of three elements: Carbon, Hydrogen, and Oxygen. The molecular weight of Glutaraldehyde is 100.1163 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements.
Sodium Acetate Trihydrate [CH3COONa.3H2O] Molecular Weight Calculation
![]()
Sodium acetate (CH3COONa) is an organic compound of four elements: Carbon, Hydrogen, Sodium, and Oxygen. The trihydrate form of Sodium acetate (CH3COONa.3H2O) also contains 3 water molecules. The molecular weight of Sodium acetate trihydrate is 136,08 which can be calculated by adding up the total weight (atomic weight multiplied by their number) of all its elements and water molecules.
ChemDB: Sodium Acetate Trihydrate (CH3COONa.3H2O)
![]()
Chemical name Sodium acetate trihydrate Chemical formula CH3COONa.3H2O Chemical nature Salt of acetic acid and sodium Molecular weight 136.08 CAS…
ChemDB: Sodium Acetate Anhydrous (CH3COONa)
![]()
Chemical name Sodium acetate (anhydrous form) Chemical formula CH3COONa Chemical nature Salt of acetic acid and sodium Molecular weight 82.03…
Suppliers: Sodium Acetate Trihydrate (CH3COONa.3H2O)
![]()
Note: Sodium acetate trihydrate (CH3COONa.3H2O) can be purchased from the following suppliers. You must visit the supplier’s product page to…
Suppliers: Sodium Acetate Anhydrous (CH3COONa)
![]()
Note: Sodium acetate anhydrous (CH3COONa) can be purchased from the following suppliers. You must visit the supplier’s product page to…
Preparation of 3 M Sodium Acetate Solution, pH 5.2 from Sodium Acetate Trihydrate (CH3COOH.3H2O)
![]()
OVERVIEWSodium acetate is a sodium salt of acetic acid which dissolves readily in water. It is commercially supplied as both…
Preparation of 3 M Sodium Acetate (CH3COONa) Solution, pH 5.2
![]()
Sodium acetate is a sodium salt of acetic acid which dissolves readily in water. It is commercially supplied as both anhydrous (molecular weight: 82.03) and trihydrate (molecular weight: 136.08) forms and both can be used to prepare 3M Sodium acetate solution. Since anhydrous sodium acetate absorbs water from the atmosphere, weighing it accurately is difficult as exposure to air can cause anhydrous sodium acetate to turn to hydrated form.
Here we show a procedure to prepare 100 ml of 3M Sodium acetate, pH 5.2 from sodium acetate anhydrous. Briefly, to prepare a 100 ml solution of 3M Sodium acetate of pH 5.2, dissolve 24.61 g Sodium acetate anhydrous in 80 ml water, adjust the pH 5.2 using acetic acid, and then make the final volume to 100 ml. Remember that this solution act as a buffer solution (Acetic acid-Sodium acetate buffer)
Sodium Acetate
![]()
Summary Chemical name Sodium acetate Chemical formula CH3COONa Chemical nature Sodium salt of acetic acid Most stable form (room temperature)…
Buffer
![]()
A buffer is an aqueous solution that can resist pH changes upon the addition of small amounts of acids and…
Topic: Dithiothreitol (DTT)
![]()
NOTES Dithiothreitol (DTT) ChemDB: Dithiothreitol (DTT) SOLUTION PREPARATION Preparation of 1 M Dithiothreitol (DTT) Solution CALCULATIONS Dithiothreitol (DTT) [C4H10O2S2] Molecular…

![Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation Sodium Phosphate Dibasic Dihydrate (Na2HPO4.2H2O) Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2023/01/fi-sodium-phosphate-dibasic-dihydrate-molecular-weight-calculation-100x77.jpg)
![Glutaraldehyde [(CH2)3(CHO)2] Molecular Weight Calculation Glutaraldehyde [(CH2)3(CHO)2] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2023/01/fi-glutaraldehyde-molecular-weight-calculation-100x81.jpg)
![Sodium Acetate Trihydrate [CH3COONa.3H2O] Molecular Weight Calculation Sodium acetate trihydrate [CH3COONa.3H2O] Molecular Weight Calculation](https://www.laboratorynotes.com/wp-content/uploads/2022/12/fi-sodium-acetate-trihydrate-molecular-weight-calculation-100x97.jpg)